Mole Ratio Lab
Purpose:
The purpose of this lab is to figure out the ratio of moles of reactants in a chemical reaction using temperature change.
Data Tables:
|
Experiment
1 2 3 4 5 6 7 |
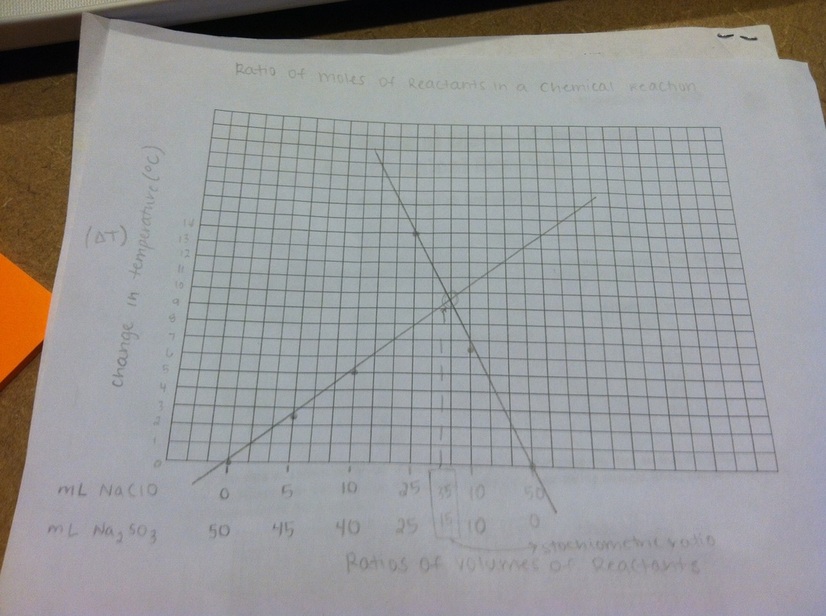
mL NaClO
0 5 10 25 40 45 50 |
mL Na2SO3
50 45 40 25 10 5 0 |
Temperatue Change (°C)
0°C 2.5°C 5.0°C 14.0°C 6.5°C 3.0°C 0°C |
Final Temperature (°C)
23.0°C 25.5°C 28.0°C 37.0°C 29.5°C 26.0°C 23.0°C |
|
Initial Temperature of NaClO (°C)
23.0°C |
Initial Temperature of Na2SO3 (°C)
23.0°C |
Graph:
Conclusion:
This lab showed that the ratio of moles of reactants in the chemical reaction of sodium hypochlorite (NaClO) and sodium sulfite (Na2SO3) is 7mL of NaClO to 3mL of Na2SO3. This was concluded because the temperature change of this reaction was the greatest at a change of 14 °C. This is the stoichiometric ratio of the equation becasue it generated heat, had a large temperature change, and was the point of intersection of the two lines of best fit.
Discussion of Theory:
The method of continuous variations was used to find the mole ratio of the reactants in this experiment. This method could be used because there were solutions with known concentration of reactants, there was a property to be measured, and because the volume of the reactants had to be kept constant. Keeping the volumes constant allowed this method to be used, because the heat generated from the reaction was proportional to the amount of reactants. This allowed the mole ratios to be found, because the volumes could be mixed a number of times to find the ratio that produced the greatest change in the property being measured. The idea of the stoichiometric ratio also allowed this method to work becasue the correct ratio will cause the largest amount of heat to be generated. Graphing the values then allows a line of best fit to be drawn, which will show a more accurate optimum ratio. The method of continuous variations could also be used to find the mole ratio of reactants by measuring the mass of precipitate formed, the color intensity of a product, or the volume of gas released. This method of continuous variation is extremely valuable in determining the mole ratio of reactants when they are not given.
Analysis Questions:
1. A constant volume of reactants had to be kept in this experiment so that the changes in temperature could be compared. The change in temperature was proportional to the amount of reactants in the chemical reaction. The heat that was generated was proportional to the size of the reaction that occurred. If the volume was not constant, the temperature change would not be proportional to the amount of reactants.
2. A limiting reagent is the reactant that will be completely used up in the reaction. It will cause the reaction to stop when it is all used up, becasue there is not any more left to react with the other reactants.
3. The measurement of temperature limited the precision of the data because the increments on the thermometer were only to the ones place. The measurement of volume had increments that measured to the tenths place on the 10mL graduated cylinder. On the 100mL graduated cylinder, the increments were larger like the thermometer. The 10mL graduated cylinder gave the most precise measurements.
4. On the upward sloping line of the graph, the NaClO is the limiting reagent. On the downward sloping line of the graph, the Na2SO3 is the limiting reagent.
5. The method of continuous variations could use other physical properties such as the mass of percipitate that is formed, the volume of gas that is released, or the color change that occurs.
6. It is more accurate to use the point of intersection of the two lines to find the mole ratio, because the point of intersection between the lines gives the stoichiometric ratio. The ratio that caused the greatest temperature change can be changed a small amount to increase the temperature change. The stoichiometric ratio does not change, and the line of best fit models the point where the ratios are just right and cannot fluctuate.
2. A limiting reagent is the reactant that will be completely used up in the reaction. It will cause the reaction to stop when it is all used up, becasue there is not any more left to react with the other reactants.
3. The measurement of temperature limited the precision of the data because the increments on the thermometer were only to the ones place. The measurement of volume had increments that measured to the tenths place on the 10mL graduated cylinder. On the 100mL graduated cylinder, the increments were larger like the thermometer. The 10mL graduated cylinder gave the most precise measurements.
4. On the upward sloping line of the graph, the NaClO is the limiting reagent. On the downward sloping line of the graph, the Na2SO3 is the limiting reagent.
5. The method of continuous variations could use other physical properties such as the mass of percipitate that is formed, the volume of gas that is released, or the color change that occurs.
6. It is more accurate to use the point of intersection of the two lines to find the mole ratio, because the point of intersection between the lines gives the stoichiometric ratio. The ratio that caused the greatest temperature change can be changed a small amount to increase the temperature change. The stoichiometric ratio does not change, and the line of best fit models the point where the ratios are just right and cannot fluctuate.