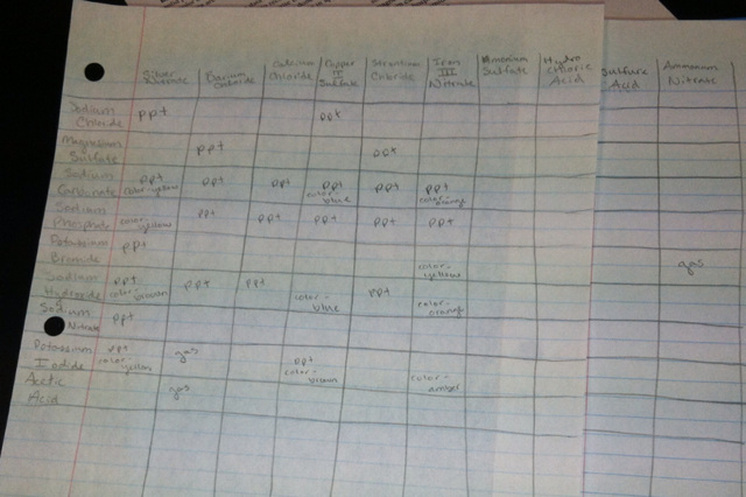
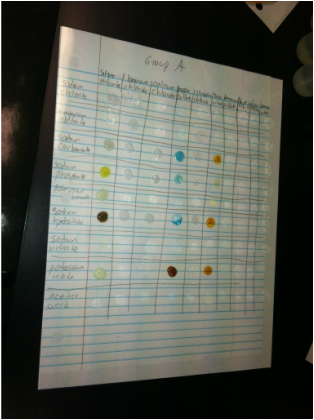
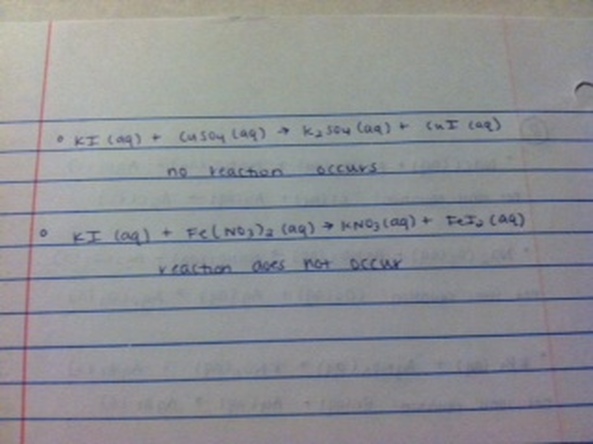Data Table:
Conclusion:
In this lab, some solutions reacted while others did not. The solutions that reacted showed signs that a reaction had occured such as color change, a percipitate formed, or a gas released.
Discussion of theory:
This lab focused on reacting chemicals to see what products, if any, would form. In this lab, there were ten group A solutions. These solutions were then mixed, one by one, with the nine group B elements. This lab allowed many reactions to be set up, and completed. The results were then observed. The results that were observed were used to study which solutions reacted with many other solutions, and which solutions did not. If a reaction does not occur, it means that when the two solutions were mixed, there was no change. The reaction produced a solution of ions that is the same as the starting solutions mixed together. Some solutions react because one of the products is insoluble. If both of the products are insoluble, the reaction will not occur. This lab required the knowledge to set up and balance chemical equations. Chemicals had to be translated from words to symbols. This aspect of the lab practiced naming compounds. Once the solutions were named, they were set up in a reaction. This part of the lab used knowledge of types of reactions. The reactants had to be switched to become products. Also, charges had to be crossed to write the equations correctly. Solubility rules were a huge part of this lab, because each solution had to be identified as aqueous, liquid, gas or solid. After the equation was set up, it had to be balanced. The balanced molecular equation then had to be made into a net ionic equation. A net ionic equation is an equation that only includes the parts of the reaction that participate in the chemical change. The ions that do not participate in the chemical change are called spectator ions. Spectator ions can be removed because they do not change chemically. The net ionic equation was found by identifying and removing the spectator ions from the equation. This allowed the solutions that reacted to be specified. Once the reactions occurred, then they could be identified using the types of metathesis reactions. The reactions could be classified as a precipitation reaction, a neutralization reaction, or a gas formation reaction. It was important in this lab to know how to set up reactions and then identify the results.
Sources of Error:
Most sources of error for this lab would be human related and random. Possible sources of error could have been accidentally using the wrong solution to be mixed, not mixing enough of the two solutions, or not making detailed observations. If the wrong solutions were mixed, the results would not be correct. If not enough of the two solutions were mixed, the reaction may not occur as obviously. This could allow the group to miss signs that a reaction occurred. Also, if very detailed observations are not taken, then the reaction may be looked over. It is important to observe closely so that nothing is missed, such as air bubles after the reaction that wold be very important to the reaction occuring.
Analysis Questions:
1. From the data collected, Silver Nitrate, Barium Chloride, Sodium Carbonate, Sodium Phosphate, Sodium Hydroxide, Potassium Iodide and Copper II Sulfate reacted with most solutions. These solutions formed percipataces and caused color changes when they were mixed with many other solutions. On the other hand, Acetic Acid, Sodium Nitrate, Potassium Bromide, Ammonium Nitrate, Sulfuric Acid, Hydrochloric Acid, Ammonium Sulfate, and Soduim Chloride reacted with almost no other solutions. When mixed, these solutions hardly reacted with other solutions.
2.
3. The types of metathesis reactions are precipitation reactions, neutralization reactions, and gas formation reactions. A precipitation metathesis reaction is a reaction that produces a precipitate. A neutralization metathesis reaction is a reaction that produces neutral water. A gas formation methathesis reacton forms a gas.
4. Many things are soluble in water because water is the universial solvent. Water is polar, and it has electron pairs on top of the molecule. This causes water to be bent, and to have a slight potive charge and a slight negative charge. These slight charges allow substances to be attracted to these charges. Water will then dissociate the compounds. This means that water will pull the parts of the compound apart. Next, water will cause hydration to occur. Hydration is when water surrounds a compound. These properies allow water to dissolve many substances. Some things do not dissolve in water becasue they are very large or because they are hydrophobic. Substances such as oil do not dissolve in water because the oil is a lipid and lipids repell water.
5. A double replacement reaction that produces two aqueous products is considered "no reaction" because when the two solutions are added together in a beaker, the ions are the same after the reaction. When the solutions start, they are ions mixed together in a beaker, and when the reaction is over they are still just a "soup" of ions in a beaker.